|
The effective dose received in a typical chest X-ray examination is about 0.05 mSv. A radiation worker may legally receive up to 20 mSv per year. The annual dose received by a member of the public in Hong Kong from natural background radiation is about 2 mSv. Caesium is also used in the manufacturing of infrared lamps. The attributes of naturally decaying atoms, known as radioisotopes, give. Radioisotopes and radiation used in food and agriculture are helping to reduce these figures. Caesium is widely used in the production of electricity, electronics and in chemistry. The United Nations (UN) Food and Agriculture Organization (FAO) estimates that about 690 million people (about one in ten) were suffering from chronic undernourishment in 2019. Caesium nitrate is used to make optical glasses. Caesium is used in industries as a catalyst promoter. The metal is used in the manufacture of photocells and in the removal of residual gases from vacuum tubes. This is a very large unit and submultiples, in particular, the millisievert (mSv - one thousandth of a sievert) are commonly used. Caesium compounds with chlorides are used in photoelectric cells. Rubidium and its salts have few commercial uses. The effective dose is frequently abbreviated to dose.  
Commercial use of the Images will be charged at a rate based on the particular use, prices on application. The effective dose is a single number broadly representing the risk to health taking into consideration the susceptibility to harm of different tissues and weighted for the harmfulness of different types of radiation. If you wish to use the Images in a manner not permitted by these terms and conditions please contact the Publishing Services Department by email. To put all ionising radiation on an equal basis with regard to the potential for causing harm, a quantity known as effective dose is introduced. 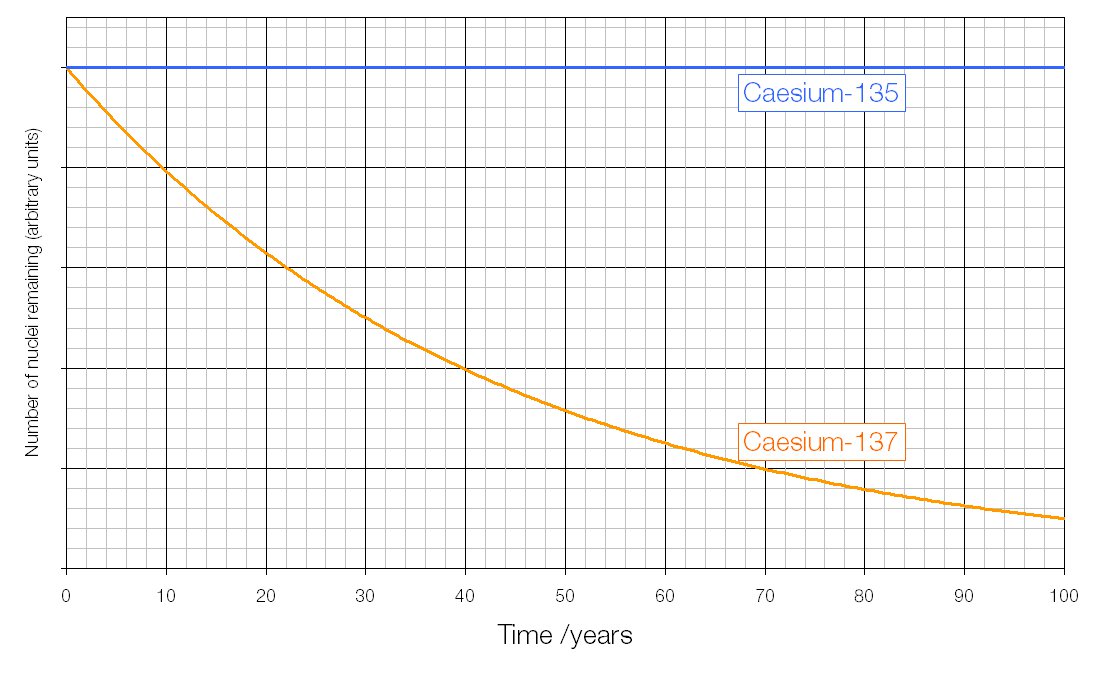
Dose and Sievert - The effect of radiation on any material is determined by the "dose" of radiation that material receives. One of the most widespread everyday uses of caesium is its application in atomic clocks. Isotopes of an element can be classified as radioisotopes (those undergoing decay with the emission of characteristic radiation) or stable isotopes. The Use of Caesium and the Atomic Clocks. Different isotopes have the same chemical properties, but somewhat different physical properties. Cesium was used in vacuum tubes to remove traces of remaining oxygen due to its ready nature to bond with it, and as a coating on heated cathodes to increase the electric current. Atoms of an element having the same number of protons but different number of neutrons in the nuclei.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
